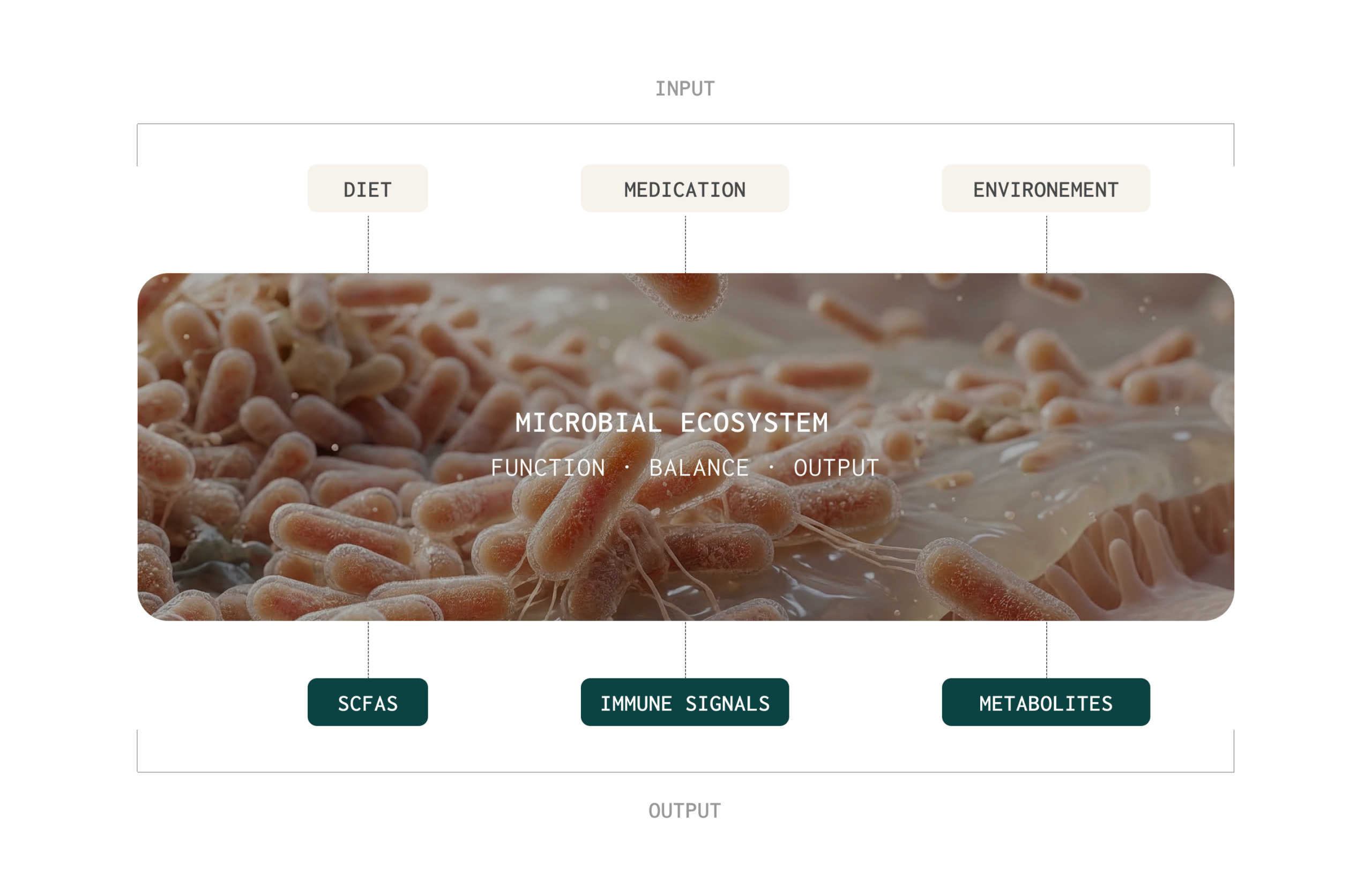
The microbiome is a dynamic ecosystem, not a collection of individual species
Inside every person lives a vast community of microorganisms — mostly bacteria, along with
fungi, viruses and archaea — collectively called the microbiome. This community is densest in
the gut, especially the colon, where trillions of microbes interact with each other and with their
human host. 1,2
Rather than acting as isolated species, these microbes form an ecosystem that adjusts to diet,
medications, infections, stress and other exposures. The functional profile of this ecosystem
depends on many moving parts: which microbes are present, how they interact, what fuels they
receive, the gut environment, and which metabolic activities are turned on at any given time. In
turn, these metabolic outputs shape host physiology and can promote health or, if out of
balance, may contribute to symptoms and disease processes.3,4
Microbial diversity is the first line of defence against pathogens
One of the most important ways the microbiome supports health is by helping protect against harmful microorganisms, a process called colonisation resistance. In a diverse, well-balanced community, many species compete for space and nutrients, leaving few open niches for potential pathogens to occupy. This competitive exclusion means that invaders struggle to find food or sites to attach and grow.
pH modulation
Production of acids and SCFAs lowers luminal pH, making the colon less hospitable for certain harmful bacteria.
Antimicrobial compounds
Generation of bacteriocins and other compounds that directly inhibit competitors.
Barrier maintenance
Support of the mucus layer and gut barrier, limiting pathogen and toxin access to the intestinal wall.
Immune priming
Promotion and priming of innate immune cells, enabling faster response to enteric infection.
When diversity is reduced — after antibiotics, severe illness or restrictive diets — this protective network can weaken. With fewer competitors and less functional redundancy (fewer microbes able to perform the same protective functions), opportunistic organisms may gain a foothold and, in some cases, establish infection or persistent colonisation.
Clinical example
Impaired colonisation resistance has been linked to greater susceptibility to post-infectious IBS following gastroenteritis, with some individuals experiencing long-term symptoms after an acute infection. 9,10

Diet determines what the microbiome produces
Fibre fermentation and health-supporting metabolites
What microbes do is strongly influenced by what they are fed. Many dietary fibres cannot be
digested by human enzymes but can be fermented by gut microbes. When bacteria ferment
complex carbohydrates, they can produce short-chain fatty acids such as acetate, propionate
and butyrate.11,12
Critically, the gut microbiota is metabolically flexible. Microbes that produce beneficial metabolites from dietary fibre can shift towards less favourable outputs when the diet is low in fermentable carbohydrates and high in protein or fat. Different fibres (resistant starch, inulin, arabinoxylans) favour different microbial groups and can shift SCFA profiles, so the type and diversity of fibre matter as much as total fibre intake.
Clinical example
Impaired colonisation resistance has been linked to greater susceptibility to post-infectious IBS following gastroenteritis, with some individuals experiencing long-term symptoms after an acute infection. 9,10

Microbial metabolites shape immune function across the lifespan
Early-life training.
From birth, the immune system learns to distinguish between threats and harmless exposures. Microbes are key teachers in this process. Early contact with a diverse set of microbes helps immune cells learn when to respond strongly and when to remain tolerant, supporting the development of regulatory pathways that reduce the risk of over-reactive responses.
Disruptions in early-life microbial exposure — frequent antibiotics, limited dietary diversity or lack of environmental microbial contact — have been associated with increased risk of allergies, asthma, inflammatory bowel disease and some autoimmune conditions later in life.
Ongoing immune modulation in adults
In adulthood, the microbiome continues to shape immune tone. Microbial products interact with pattern-recognition receptors and other immune sensors, sending signals that can either amplify or dampen inflammation.
When the microbiome is balanced, these signals usually support a steady, controlled immune state. When microbial composition and function are disrupted, signalling may become skewed in ways that can contribute to chronic inflammation in susceptible individuals.24
Clinical example
In oncology, gut microbiome composition has been linked to better or worse responses to
immunotherapy, suggesting microbial immune modulation can influence treatment outcomes.
Health outcomes depend on microbial cooperation
Cross-feeding and microbial cooperation
Many clinically relevant metabolites are not produced by a single microbe acting alone. Instead, they arise through cross-feeding, where one species’ metabolic by-products become another species’ fuel. For example, fibre fermentation by Bifidobacterium spp. can generate acetate and lactate that is then converted into butyrate by butyrate-producing Firmicutes such as Faecalibacterium prausnitzii, Roseburia and Eubacterium.28

This distributed architecture also helps explain how microbial communities become more stable and resilient to disturbances, and how people with very different species profiles can still display similar functional capacities: different microbial “teams” can lead to similar metabolic outcomes.
 |
 |
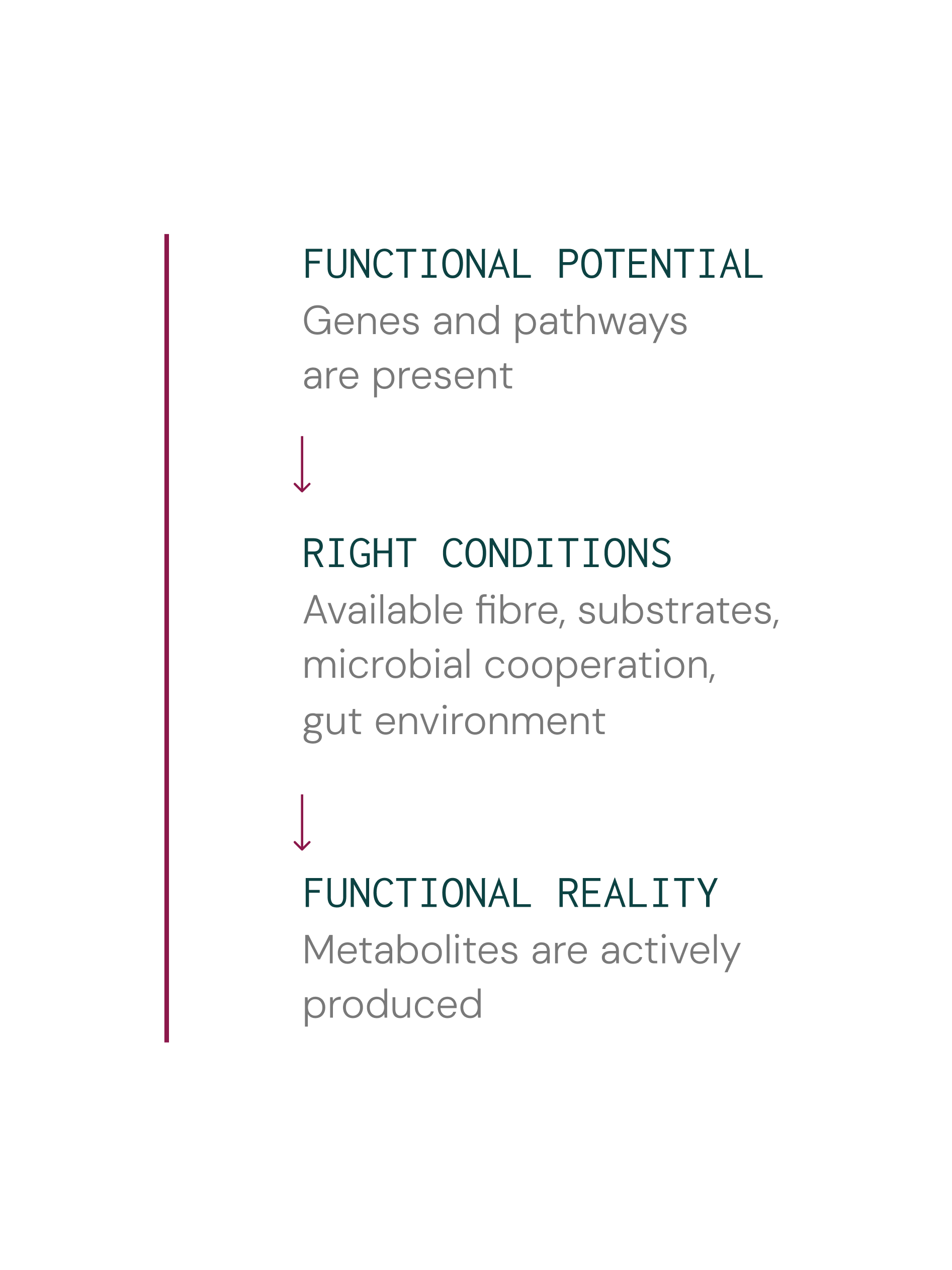
Functional potential only becomes
functional reality when the right conditions align
Modern sequencing technologies can estimate the microbiome’s functional potential by identifying genes and pathways — a valuable starting point. However, this potential must be realised through the right conditions, and especially fuel source availability.
What bacteria produce depends heavily on available substrates: when fermentable carbohydrates are plentiful, SCFA production often increases; but when dietary fibre is limited, proteolytic fermentation pathways can become more prominent.11
CLINICAL EXAMPLE
Individuals with higher butyrate-producing capacity may show greater response and metabolic benefit when diets provide matching fermentable fibres, illustrating how diet can “unlock” functional capacity.31,32,33

Health as a functional balance
Because healthy people can have very different species profiles, it is difficult to define a single "ideal" microbiome based only on who is present. It is more practical to think in terms of functional balance.34
Functional balance
Functional dysbiosis
A functional microbiome lens helps explain why the same intervention — higher fibre intake, probiotics or medication — may help one person but worsen symptoms in another, depending on microbial functions, dietary context and host factors. Supporting health often means working with the ecosystem: providing the right fuels, avoiding unnecessary disruptions, and using targeted strategies to shift microbial functions rather than chasing or eradicating individual species.

Key messages
Key POINT
The microbiome is a dynamic ecosystem whose functions — not just its composition — shape human health. Diverse communities support colonisation resistance, diet determines metabolic outputs, and health outcomes depend on community cooperation and functional balance rather than individual species.1,3