The microbiome is a dynamic ecosystem, not a collection of individual species
Every person harbours a vast community of microorganisms — mostly bacteria, along with fungi, viruses and archaea — collectively known as the microbiome. This community is densest in the gut, especially the colon, where trillions of microbes interact with each other and with their human host. Rather than acting as isolated species, these microbes form an ecosystem that adjusts to diet, medications, infections, stress and other exposures. 1,2,3
The functional outputs of this ecosystem depends on factors such as which microbes are present, how they interact, what fuels they receive, the gut environment, and which metabolic activities are turned on at any given time.1,2,3
Microbial diversity protects against pathogens through colonisation resistance
One of the microbiome’s key roles is protecting against harmful microorganisms, a process called colonisation resistance. In a diverse, well-balanced community, many species compete for space and nutrients, leaving few open niches for potential pathogens to occupy. Other mechanisms of colonisation resistance include the production of acidic metabolites that lower luminal pH, antimicrobial compounds, and metabolites that support barrier function and host immunity.4,5,6,7
Did you know?
When diversity is reduced — after antibiotics, severe illness or restrictive diets — this protective network can weaken. With fewer competitors and less functional redundancy (fewer microbes able to perform the same protective functions), opportunistic organisms may gain a foothold and, in some cases, establish infection or persistent colonisation.4,7
Clinical example
Impaired colonisation resistance has been linked to greater susceptibility to post-infectious IBS following gastroenteritis, with some individuals experiencing long-term symptoms after an acute infection. 8,9

Diet determines microbial outputs
The same microbes can produce different metabolites
What microbes do is strongly influenced by what they are fed. Many dietary fibres cannot be digested by human enzymes but can be fermented by gut microbes, producing health-promoting short-chain fatty acids (SCFAs). 10,11 Microbes also ferment undigested protein and amino acids in the distal colon, producing metabolites that may be harmful or beneficial. 10

Critically, the gut microbiota is metabolically flexible. Microbes that produce beneficial metabolites from dietary fibre can shift towards less favourable outputs when the diet is low in fermentable carbohydrates and high in protein or fat. 10,13
The type and diversity of fibre matter as much as total fibre.
Microbial metabolites shape immune
function across the lifespan
From birth, the immune system learns to distinguish between threats and harmless exposures. Early contact with diverse microbes supports the development of regulatory pathways that reduce the risk of over-reactive responses. Disruptions in early-life microbial exposure have been associated with increased risk of allergies, asthma, inflammatory bowel disease and autoimmune conditions.21
In adulthood, the microbiome continues to shape immune function: Microbiota-derived SCFAs can promote regulatory immune responses. 14,24 Similarly, tryptophan-derived metabolites can modulate innate immune responses. 25 Lipopolysaccharides (bacterial cell wall components) can either trigger or dampen inflammatory signalling. 26 When the microbiome is balanced, these signals usually support a steady, controlled immune state. 27
Clinical example
In oncology, gut microbiome composition has been linked to better or worse responses to immunotherapy, suggesting microbial immune modulation can influence treatment outcomes.28
Health outcomes depend on community cooperation, not individual organisms
Many clinically relevant metabolites are not produced by a single microbe acting alone. Instead, they arise through cross-feeding, where one species’ metabolic by-products become another species’ fuel. People with very different species profiles can still display similar functional capacities: different microbial “teams” can lead to similar metabolic outcomes.3,29
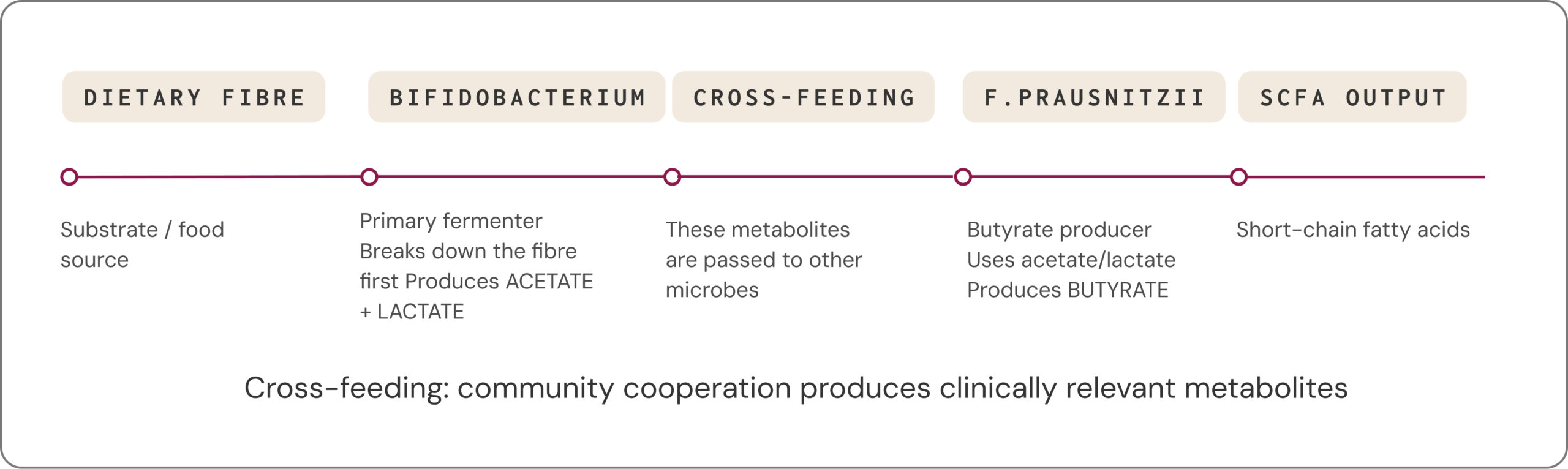
Clinical example
After antibiotic courses, some individuals develop prolonged bloating, pain and altered bowel habits. Studies have reported disrupted cross-feeding networks and lower SCFA production in these settings, consistent with loss of key metabolic partners and reduced ecosystem resilience.30

Functional potential only becomes functional reality when the right conditions align
Modern sequencing technologies can estimate the microbiome’s functional potential by identifying genes and pathways — a valuable starting point. However, this potential must be realised through the right conditions, and especially fuel source availability.3
Because healthy people can have very different species profiles, it is difficult to define a single “ideal” microbiome based only on who is present.
It is more practical to think in terms of functional balance: a state where activities that support health are robust, while pathways that generate potentially harmful metabolites are kept in check.31
FUNCTIONAL POTENTIAL
Genes and pathways are present
RIGHT CONDITIONS
Available fibre, substrates, microbial cooperation, gut environment
FUNCTIONAL REALITY
Metabolites are actively produced
Key Takeaway
The microbiome is a dynamic ecosystem whose functions — not just its composition — shape human health. Diverse communities support colonisation resistance, diet along with other factors determine metabolic outputs, and health outcomes depend on community cooperation and functional balance rather than individual species.
Functional potential only becomes functional reality when the right fuels and conditions align. A functional, ecosystem-based lens supports more personalised, targeted strategies to maintain or restore health.
- Coyte KZ, Schluter J, Foster KR. The ecology of the microbiome: networks, competition, and stability. Science. 2015;350:663–666.
- Schroeder BO, Bäckhed F. Signals from the gut microbiota to distant organs in physiology and disease. Nat Med. 2016;22:1079–1089.
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486:207–214.
- Sorbara MT, Pamer EG. Interbacterial mechanisms of colonization resistance. Mucosal Immunol. 2019;12:1–9.
- Caballero-Flores G, Pickard JM, Núñez G. Microbiota-mediated colonization resistance. Nat Rev Microbiol. 2023;21:347–360.
- Kamada N, Chen GY, Inohara N, Núñez G. Control of pathogens and pathobionts by the gut microbiota. Nat Immunol. 2013;14:685–690.
- Buffie CG, Pamer EG. Microbiota-mediated colonization resistance against intestinal pathogens. Nat Rev Immunol. 2013;13:790–801.
- Jalanka J, Gunn D, Singh G, Krishnasamy S, Lingaya M, Crispie F, et al. Postinfective bowel dysfunction following Campylobacter enteritis. Gut. 2023;72:451–459.
- Marshall JK, Thabane M, Garg AX, Clark WF, Moayyedi P, Collins SM. Eight year prognosis of postinfectious IBS. Gut. 2010;59:605–611.
- Rowland I, Gibson G, Heinken A, Scott K, Swann J, Thiele I, et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur J Nutr. 2018;57:1–24.
- Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F. From dietary fiber to host physiology: SCFAs as key bacterial metabolites. Cell. 2016;165:1332–1345.
- Peng L, Li ZR, Green RS, Holzman IR, Lin J. Butyrate enhances the intestinal barrier. J Nutr. 2009;139:1619–1625.
- Gilbert MS, Ijssennagger N, Kies AK, van Mil SWC. Protein fermentation in the gut. Am J Physiol Gastrointest Liver Physiol. 2018;315:G159–G170.
- Corrêa-Oliveira R, Fachi JL, Vieira A, Sato FT, Vinolo MAR. Regulation of immune cell function by short-chain fatty acids. Clin Transl Immunol. 2016;5:e73.
- Alexeev EE, Lanis JM, Kao DJ, Campbell EL, Kelly CJ, Battista KD, et al. Microbiota-derived indole metabolites promote intestinal homeostasis through regulation of interleukin-10 receptor. Am J Pathol. 2018;188:1183–1194.
- Canfora EE, Jocken JW, Blaak EE. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat Rev Endocrinol. 2015;11:577–591.
- Sinha AK, Laursen MF, Brinck JE, Rybtke ML, Hjørne AP, Procházková N, et al. Dietary fibre directs microbial tryptophan metabolism via metabolic interactions in the gut microbiota. Nat Microbiol. 2024;9:1964–1978.
- Teichmann J, Cockburn DW. Butyrate production dependent on resistant starch source. Front Microbiol. 2021;12:640253.
- Venkataraman A, Sieber JR, Schmidt AW, Waldron C, Theis KR, Schmidt TM. Variable responses to dietary supplementation with resistant starch. Microbiome. 2016;4:33. 20. Ďásková N, Modos I, Krbcová M, Kuzma M, Pelantová H, Hradecký J, et al. Multi-omics signatures predict metabolic response to dietary inulin. Nutr Diabetes. 2023;13:7.
- Gensollen T, Iyer SS, Kasper DL, Blumberg RS. How colonization by microbiota in early life shapes the immune system. Science. 2016;352:539–544.
- Wernroth M-L, Fall K, Svennblad B, Ludvigsson JF, Sjölander A, Almqvist C, et al. Early childhood antibiotic treatment is associated with risk of type 1 diabetes. Diabetes Care. 2020;43:991–999.
- Kronman MP, Zaoutis TE, Haynes K, Feng R, Coffin SE. Antibiotic exposure and IBD development among children. Pediatrics. 2012;130:e794–e803.
- Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. 2013;504:451–455.
- Zelante T, Iannitti RG, Cunha C, De Luca A, Giovannini G, Pieraccini G, et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor. Immunity. 2013;39:372–385.
- Mohr AE, Crawford M, Jasbi P, Fessler S, Sweazea KL. Lipopolysaccharide and the gut microbiota: considering structural variation. FEBS Lett. 2022;596:849–875.
- Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020;30:492–506.
- Baruch EN, Youngster I, Ben-Betzalel G, Ortenberg R, Lahat A, Katz L, et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma. Science. 2021;371:602–609.
- Culp EJ, Goodman AL. Cross-feeding in the gut microbiome. Cell Host Microbe. 2023;31:485–499.
- Palleja A, Mikkelsen KH, Forslund SK, Kashani A, Allin KH, Nielsen T, et al. Recovery of gut microbiota following antibiotic exposure. Nat Microbiol. 2018;3:1255–1265.
- Tiffany CR, Bäumler AJ. Dysbiosis: from fiction to function. Am J Physiol Gastrointest Liver Physiol. 2019;317:G602–G608.