The microbiome is a critical organ in health, not a wellness trend
The gut microbiome influences key functions such as metabolism, immune regulation, gut barrier function, and systemic signalling. This makes it an important consideration in clinical practice, especially in patients with ongoing symptoms or complex chronic conditions.1
Microbiome research is no longer about detection, it’s about function
Over the past two decades, microbiome research has moved from simply identifying which microbes are present to understanding what they do and how they interact with the host.2 Large population studies have shown that while microbial species differ widely between individuals, many core metabolic functions are preserved.3
EARLY 2000S
Species cataloguing and taxonomic classification
2010’s
Functional profiling and metagenomic sequencing
current
Clinical integration and longitudinal intervention studies
Advances in metagenomic sequencing now enable functional profiling — assessing microbial gene content and pathway capacity rather than relying on species-level associations alone.3 Longitudinal studies and controlled human interventions continue to clarify how the microbiome responds to diet, medication and environmental exposure.4,5
Microbial function significantly
influences chronic conditions
The gut microbiome can influence chronic disease by affecting core functions such as metabolism, immune regulation, gut barrier integrity, and systemic signalling.1 This helps explain why microbial imbalance may contribute to both gastrointestinal symptoms and wider systemic presentations. 1,6,7,8
In practice, these disturbances may present as persistent inflammatory symptoms, heightened sensitivity to dietary triggers, altered bowel habits, fatigue, or variable response to established treatments.1,6,7
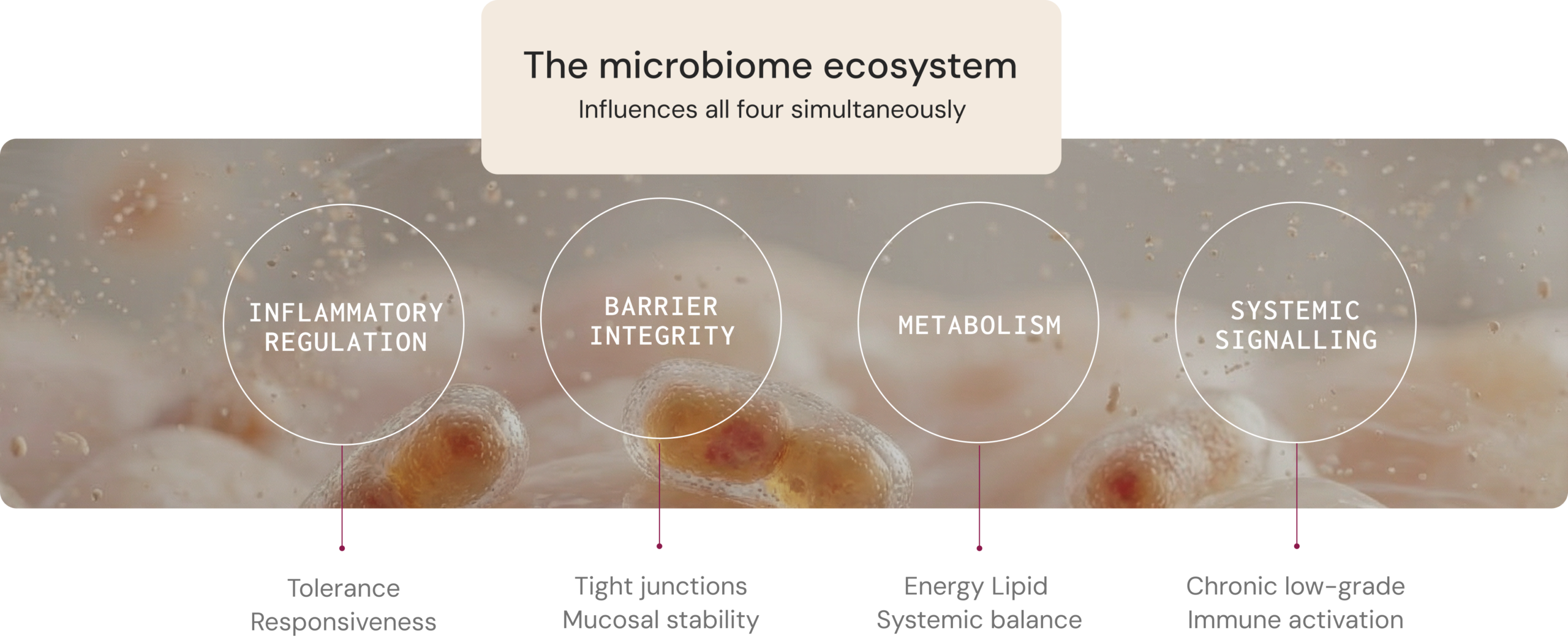
Four core physiological functions
influenced by the microbiome
The microbiome influences health through measurable host–microbe interactions that affect metabolism, immune regulation, gut barrier integrity, and systemic signalling. Understanding these mechanisms can help clinicians interpret how microbial ecology may contribute to symptom patterns and disease progression.1
Immune
regulation
Microbial components and metabolites interact continuously with immune cells within the intestinal mucosa. Short-chain fatty acids and other microbial metabolites influence regulatory T-cell activity, cytokine production and immune signalling pathways.6
IBD9, type 1 diabetes10, rheumatoid arthritis10
Gut barrier
function
Microbial metabolites support epithelial energy metabolism, tight junction stability, and mucosal signalling. When disrupted, increased permeability and altered mucosal signalling may contribute to immune activation and gastrointestinal symptoms.7,11
IBS12, IBD9, coeliac disease7
Metabolism
Microbial metabolites interact with host receptors involved in glucose and appetite regulation, lipid metabolism and inflammatory signalling pathways. These compounds influence endocrine signalling, energy metabolism and inflammation.8
Obesity, type 2 diabetes, NAFLD1
Systemic
signalling
Microbial metabolites interact with host receptors and influence signalling pathways beyond the gut, including enteroendocrine signalling, neural circuits, and immune pathways.11
Cardiovascular disease, Parkinson’s disease, type 2 diabetes13,14
Simplified microbiome models are not sufficient for effective care
Wellness framing
“Good vs bad bacteria.” Rebalance with a single product.
One-size-fits-all interventions. No consideration of individual ecology or patient context.
Clinical science
Dynamic ecosystems. Functional capacity and ecological structure. Strain-level differences. Context-dependent metabolic effects. Bidirectional microbiome signalling.
Two individuals taking the same probiotic or dietary intervention may experience different outcomes depending on their existing microbial ecology, metabolic pathways and host physiology.15 Without understanding this context, interventions may fail to address the mechanisms contributing to symptoms or disease progression.
A clinically responsible framework recognises individual variation, strain-level functional differences, context-dependent metabolic effects and bidirectional host–microbe signalling.2,3,15,16
Key Takeaway
The microbiome is directly involved in physiological functions central to chronic disease, including metabolism, immune regulation, gut barrier integrity, and systemic signalling.1 It is a biologically active system embedded within human physiology.
Understanding microbial ecosystem function is therefore an important component of modern clinical medicine and interpreting how the microbiome influences human health.17

- Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol. 2021;19(1):55–71. https://doi.org/10.1038/s41579-020-0433-9
- Costello EK, Stagaman K, Dethlefsen L, Bohannan BJ, Relman DA. The application of ecological theory toward an understanding of the human microbiome. Science. 2012;336(6086):1255–62.
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486(7402):207–14.
- Dethlefsen L, Relman DA. Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc Natl Acad Sci USA. 2011;108(Suppl 1):4554–61.
- Zhou X, Shen X, Johnson JS, et al. Longitudinal profiling of the microbiome at four body sites reveals core stability and individualized dynamics during health and disease. Cell Host Microbe. 2024;32(4):506–526.e9.
- Tan J, McKenzie C, Potamitis M, Thorburn AN, Mackay CR, Macia L. The role of short-chain fatty acids in health and disease. Adv Immunol. 2014;121:91–119.
- Ornelas A, Dowdell AS, Lee JS, Colgan SP. Microbial Metabolite Regulation of Epithelial Cell-Cell Interactions and Barrier Function. Cells. 2022;11(6):944.
- Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell. 2016;165(6):1332–1345. https://doi.org/10.1016/j.cell.2016.05.041
- Lloyd-Price J, Arze C, Ananthakrishnan AN et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature. 2019;569(7758):655–662. https://doi.org/10.1038/s41586-019-1237-9
- Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature. 2016;535(7610):75–84. https://doi.org/10.1038/nature18848
- Carabotti M, Scirocco A, Maselli MA, Severi C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Ann Gastroenterol. 2015;28(2):203–209.
- Staudacher HM, Mikocka-Walus A, Ford AC. Common mental disorders in irritable bowel syndrome: pathophysiology, management, and considerations for future randomised controlled trials. Lancet Gastroenterol Hepatol. 2021;6(5):401–410. https://doi.org/10.1016/S2468-1253(20)30363-0
- de Vos WM, Tilg H, Van Hul M, Cani PD. Gut microbiome and health: mechanistic insights. Gut. 2022;71(5):1020–1032. https://doi.org/10.1136/gutjnl-2021-326789
- Cryan JF, O’Riordan KJ, Cowan CSM, et al. The microbiota-gut-brain axis. Physiol Rev. 2019;99(4):1877–2013. https://doi.org/10.1152/physrev.00018.2018
- Leshem A, Segal E, Elinav E. The Gut Microbiome and Individual-Specific Responses to Diet. mSystems. 2020;5(5):e00665-20.
- Zmora N, Suez J, Elinav E. You are what you eat: diet, health and the gut microbiota. Nat Rev Gastroenterol Hepatol. 2019;16(1):35–56. https://doi.org/10.1038/s41575-018-0061-2
- Lynch SV, Pedersen O. The human intestinal microbiome in health and disease. N Engl J Med. 2016;375(24):2369–2379. https://doi.org/10.1056/NEJMra1600266