The microbiome is a critical organ in health, not a wellness trend
The gut microbiome is a biologically active ecosystem that interfaces with core physiological functions such as metabolism, immune regulation, gut barrier integrity, and systemic signalling — functions relevant to chronic gastrointestinal and systemic disease.1
In practice, the question is rarely “is a microbe present?” It is what pattern of microbial functions or host-microbe interactions could be contributing to symptoms, inflammatory burden, barrier disruption or variable treatment response?
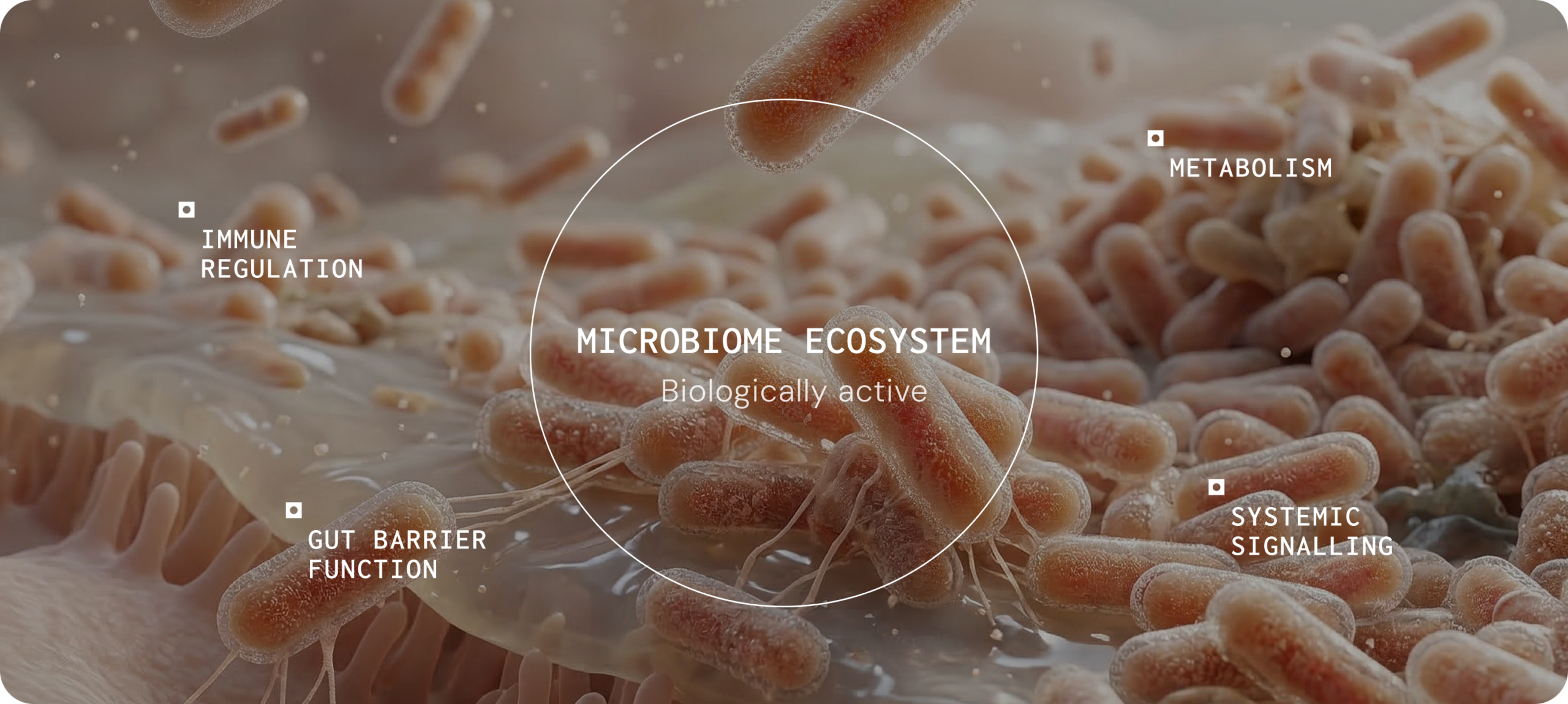
When microbial activity influences these systems, it can contribute to symptom persistence, inflammatory burden and variability in treatment response. In practice, clinicians may therefore consider whether host–microbe interactions are influencing a patient’s presentation, particularly in complex or chronic conditions where symptoms cannot be fully explained by structural pathology alone.
Integrated across core disciplines
Immunology
Immune education, inflammatory regulation, mucosal immunity
Gastroenterology
Barrier integrity, mucosal immune responses, motility
Endocrinology
Hormone regulation, appetite signalling, glucose metabolism
Metabolic medicine
Energy metabolism, lipid metabolism, systemic inflammatory tone
Positioning the microbiome within clinical medicine also requires adherence to the evidentiary and regulatory standards that govern medical practice. Clinical claims must be supported by reproducible evidence and remain proportionate to what current data can demonstrate. This is why microbiome research in medical contexts avoids overstating conclusions and remains grounded in established biological mechanisms.
Recognising the microbiome as clinically relevant does not mean attributing disease to microbial imbalance alone. Rather, it represents an additional physiological layer that interacts with systems already central to clinical medicine.
This is where microbiome science becomes clinically relevant. When microbial activity influences these systems, it can contribute to symptom persistence, inflammatory burden and variability in treatment response.
In practice, clinicians may therefore consider whether host–microbe interactions are influencing a patient’s presentation, particularly in complex or chronic conditions where symptoms cannot be fully explained by structural pathology alone.
Microbiome research is no longer about detection, it’s about function
Over the past two decades, microbiome research has moved from simply identifying which microbes are present to understanding what they do and how they interact with the body. Large population studies have shown that while microbial species differ widely between individuals, many core metabolic functions are preserved.2,3
EARLY 2000S
Species cataloguing and taxonomic classification
2010’s
Functional profiling and metagenomic sequencing
current
Clinical integration and longitudinal intervention studies
Early Microbiome Research
- Focused on identifying which microbial species are present
- Relied on organism detection and taxonomic classification
- Highlighted large species differences between individuals
- Limited ability to assess microbial function
- Mostly cross-sectional studies describing composition
Current Microbiome Research
- Focuses on what microbes do and how they interact with physiology
- Examines microbial metabolic activity and functional pathways
- Shows core metabolic functions are conserved across populations
- Metagenomic sequencing enables functional profiling
- Longitudinal studies examine response to diet and medication
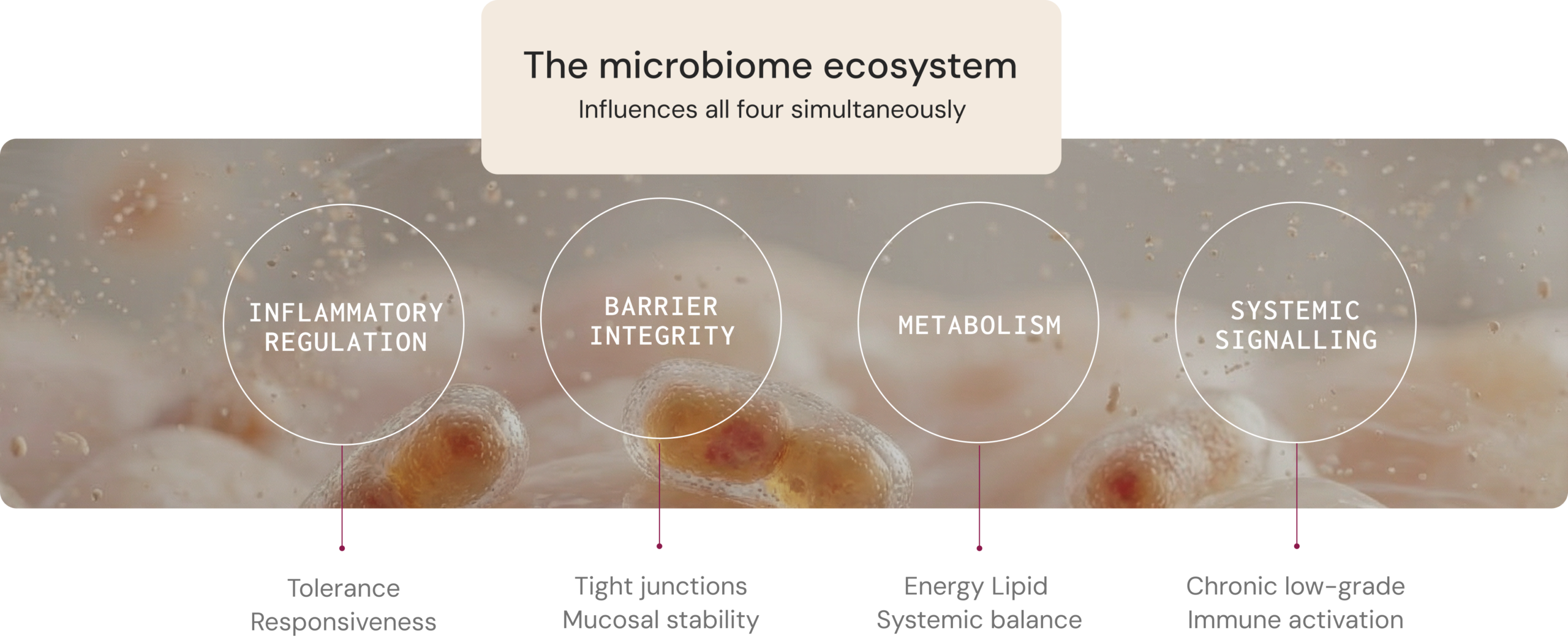
Core physiological functions influenced by the microbiome
The microbiome influences health through measurable host–microbe interactions that affect metabolism, immune regulation, gut barrier integrity, and systemic signalling. Understanding these mechanisms can help clinicians interpret how microbial ecology may contribute to symptom patterns and disease progression.1
Microbial components and metabolites interact continuously with immune cells within the intestinal mucosa, including gut-associated lymphoid tissue. Short-chain fatty acids and other microbial metabolites influence regulatory T-cell activity, cytokine production and immune signalling pathways.8
Microbial signals support immune tolerance to dietary antigens and commensal microbes while preventing excessive inflammatory activation.
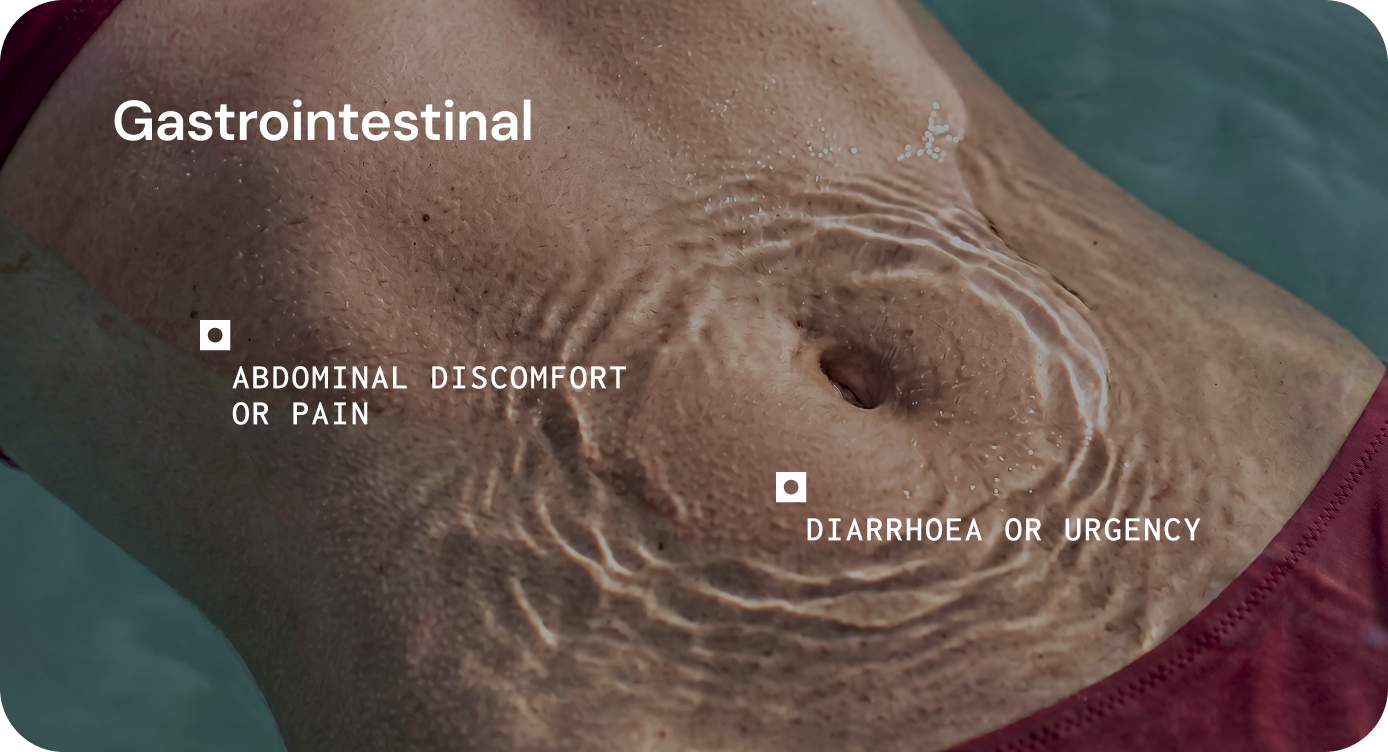
Persistent inflammatory symptoms, heightened immune sensitivity to dietary triggers, mucosal inflammation, exacerbation of immune-mediated gastrointestinal disorders.
Associated conditions
BD14, type 1 diabetes10, rheumatoid arthritis10
The intestinal epithelium is metabolically active and responsive to microbial signals. Microbial metabolites support epithelial energy metabolism, tight junction stability, and mucosal signalling, helping maintain a stable interface between luminal microbes and host tissues. When this regulation is disrupted, increased permeability and altered mucosal signalling may contribute to immune activation and gastrointestinal symptoms.9
The gut barrier functions as a stable interface between luminal microbes and host tissues, allowing efficient nutrient absorption while limiting inappropriate immune activation.
Increased intestinal permeability, impaired epithelial integrity, and altered mucosal signalling, which may contribute to inappropriate immune activation and gastrointestinal symptoms.
Associated conditions
IBS3, IBD10, coeliac disease10
Microbial metabolites interact with host receptors and influence signalling beyond the gut, including enteroendocrine signalling, neural circuits, and immune pathways. The gut and brain communicate through nerves, hormones, and immune signals. Gut microbes can influence these pathways by producing or modifying neuroactive compounds and by modulating inflammation levels.11,12
Coordinated neuroimmune and enteroendocrine signalling, stable gut–brain communication, balanced stress response.
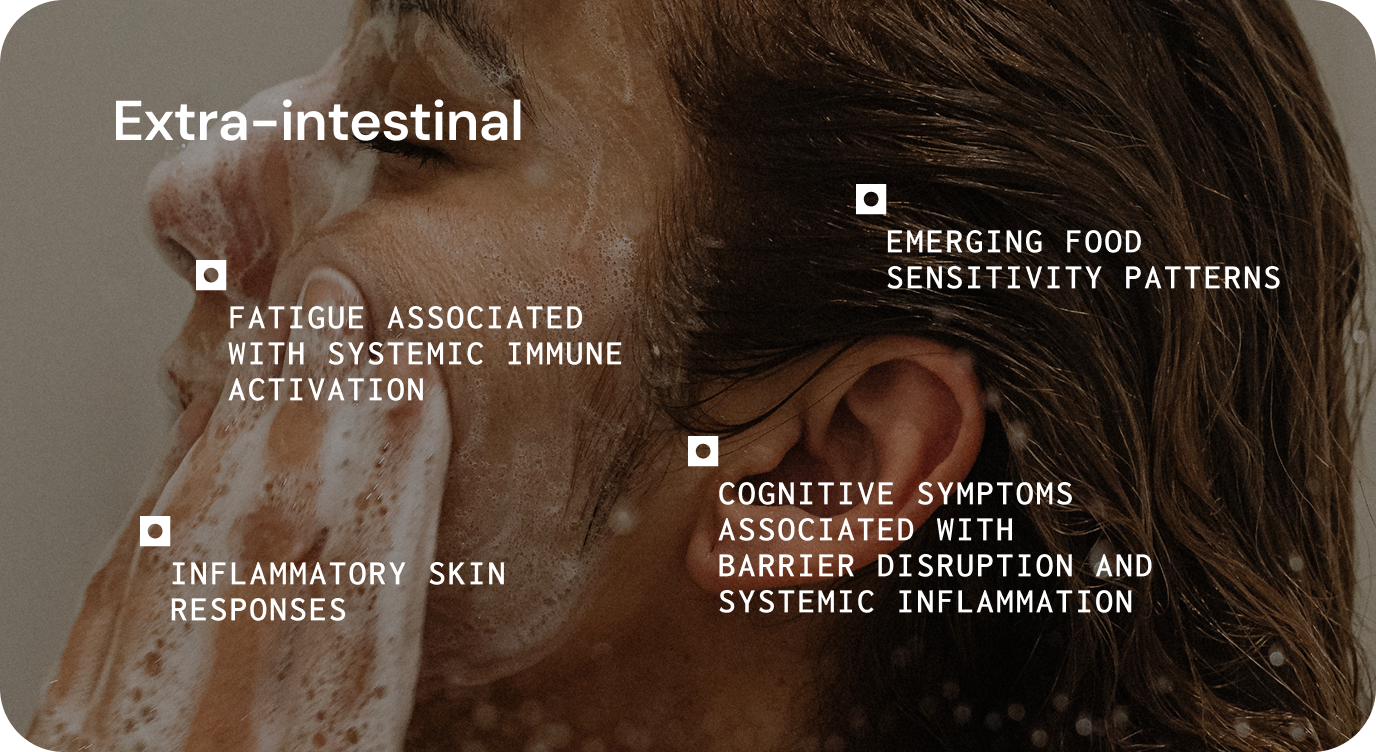
Altered stress response, neuroinflammation, disrupted gut–brain communication, variable systemic signalling.
Associated conditions
Cardiovascular disease, type 2 diabetes, Parkinson’s disease6,10
Microbial metabolites interact with host receptors involved in glucose and appetite regulation, lipid metabolism and inflammatory signalling pathways. Short-chain fatty acids and related compounds influence endocrine signalling, energy metabolism and inflammation.12
Stable energy metabolism, balanced inflammatory responses, appropriate appetite and glucose regulation.
Metabolic dysregulation, fatigue associated with inflammatory activity, altered appetite regulation, variable response to dietary or metabolic interventions.
Associated conditions
Obesity, type 2 diabetes, NAFLD3
Physiological Function Summary
Immune regulation
Immune tolerance, controlled inflammatory responses
Inflammatory symptoms, altered immune sensitivity
Barrier integrity
Stable digestion, selective permeability
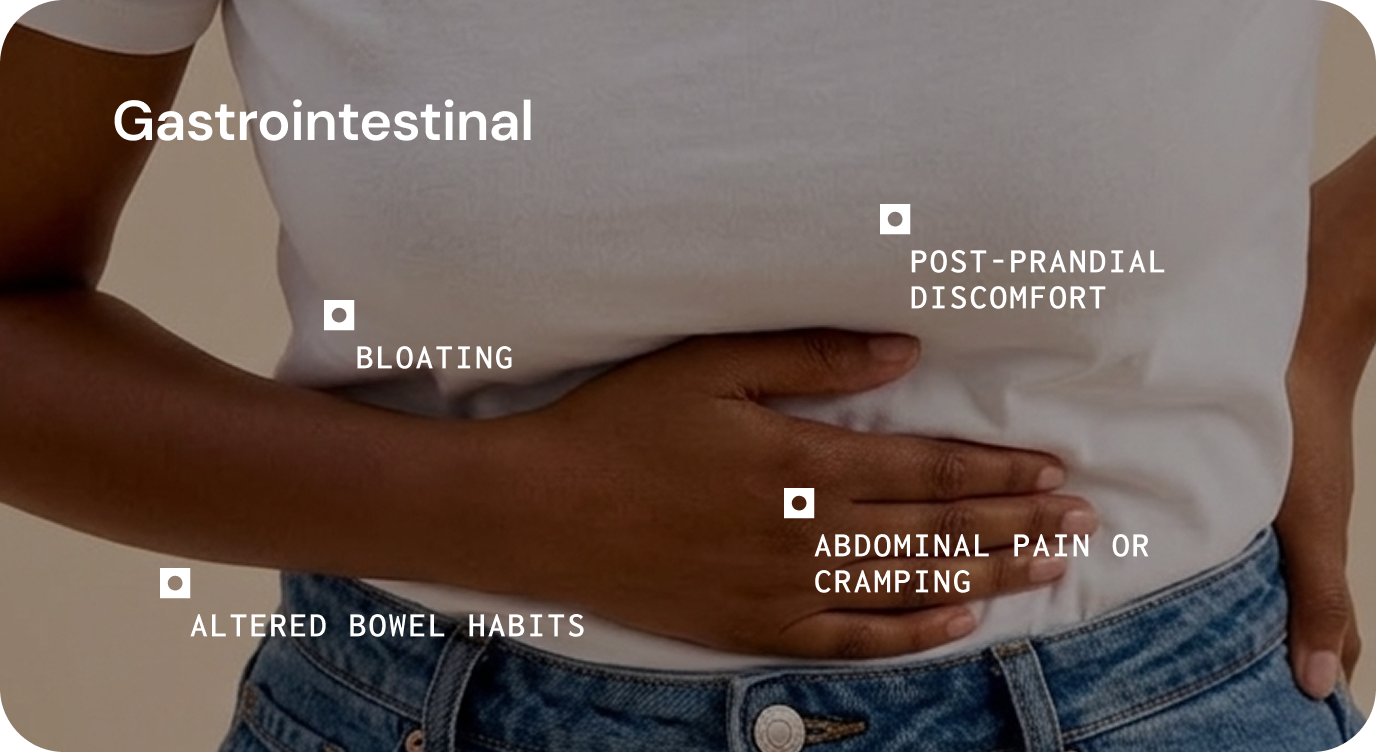
Bloating, bowel disruption, food sensitivity
Metabolic signalling
Stable energy metabolism, glucose regulation
Metabolic dysregulation, variable treatment response
Systemic signalling
Coordinated neuroimmune and enteroendocrine signalling
Disrupted gut–brain communication, variable systemic signalling
The microbiome influences chronic disease through core physiological functions
Chronic diseases rarely arise from disruption of a single biological pathway. Instead, they typically involve dysregulation across interconnected physiological functions, including metabolism, immune regulation, gut barrier integrity, and systemic signalling.1
In clinical practice, dysbiosis should not be interpreted simply as the presence of a pathogen or depletion of a single “beneficial” organism. More commonly, dysbiosis reflects broader shifts in microbial ecology and metabolic output that interact with existing clinical vulnerabilities.
What this looks like in practice
inflammatory regultion
Microbial metabolites, including short-chain fatty acids (SCFAs) and some indole derivatives, play an important role in immune regulation. These compounds influence immune cell differentiation, cytokine production and inflammatory signalling.8 Reduced SCFA-producing capacity has been associated with conditions characterised by immune dysregulation, including inflammatory bowel disease, autoimmune disorders and the low-grade systemic inflammation observed in metabolic syndrome.5,8
Clinical presentations
The following clinical presentations have been associated with microbiome-related immune dysregulation. Causal relationships are not established for all; these associations should be interpreted in the context of each patient’s clinical picture.1,8
Barrier integrity
The intestinal epithelium forms a critical interface between luminal microbial communities and the host immune system. Microbial metabolites such as butyrate support epithelial energy metabolism, tight junction integrity and mucosal immune signalling.9 When epithelial barrier regulation is disrupted, microbial products such as hexa-acylated lipopolysaccharides (hexa-LPS) may cross the intestinal barrier more readily, promoting immune activation and inflammatory signalling.1,9
Clinical presentations
The following clinical presentations have been associated with microbial disruption in the literature.
Causal relationships are not established for all; these associations should be interpreted in the context of each patient’s clinical picture
Metabolism
Gut microbes participate in nutrient metabolism and energy regulation through the production of bioactive metabolites that interact with metabolic pathways. Alterations in microbial metabolic activity have been associated with differences in glycaemic control, energy homeostasis and systemic inflammatory tone.12
Clinical presentations 1,13
Systemic signalling
Microbial products can influence signalling beyond the gut through several recognised routes, including enteroendocrine signalling, gut-innervating afferent nerves such as the vagus nerve, and immune pathways.1,13 Short-chain fatty acids, tryptophan-derived metabolites, and hexa-acylated lipopolysaccharides can act on enteroendocrine and immune cells, modulate cytokine signalling, and influence neuronal communication between the gut and brain.13
These interactions are bidirectional. Factors such as diet, immune status, medications and environmental exposures continuously shape microbial ecology, while microbial metabolites in turn influence immune regulation, gut barrier integrity, metabolism and systemic signalling. Patients with similar diagnoses can respond differently to the same interventions — differences in microbial metabolism are increasingly recognised as a contributing factor.1,8,9
Did you know?
These interactions are bidirectional. Diet, immune status, medication exposure and environmental factors continuously shape microbial ecology, while microbial metabolites in turn influence immune function, epithelial integrity and metabolic regulation. Patients with similar diagnoses can respond differently to the same interventions — differences in microbial metabolism are increasingly recognised as a contributing factor.8,9,10
Simplified microbiome models are not sufficient for clinical practice
Wellness messaging often reduces the microbiome to simplified concepts such as “good versus bad bacteria” or the idea that gut health can be restored by simply “rebalancing” microbial populations. These narratives are appealing because they are easy to communicate, but they do not reflect how microbial ecosystems function within human physiology.
Clinical science approaches the microbiome differently. Rather than focusing on individual organisms classified as beneficial or harmful, it considers microbial communities as dynamic ecosystems whose effects depend on ecological structure, functional capacity and host context.3
Wellness framing
“Good vs bad bacteria.” Rebalance with a single product.
One-size-fits-all interventions. No consideration of individual ecology or patient context.
Clinical science
Dynamic ecosystems. Functional capacity and ecological structure. Strain-level differences. Context-dependent metabolic effects. Bidirectional microbiome signalling.
This approach also reflects the clinical and regulatory standards that govern medical practice. In medical contexts, health claims must be supported by robust scientific evidence, which is why clinical microbiome research avoids overstating conclusions and remains grounded in established biological mechanisms.
As a result, interventions based purely on simplified microbial classifications may produce inconsistent outcomes. Two individuals taking the same probiotic, dietary intervention or supplement may experience different effects depending on their existing microbial ecology, metabolic pathways and host physiology.16
When microbiome interventions are selected without consideration of the clinical presentation, they may not target the biological mechanisms contributing to a patient’s symptoms. In some cases, this may result in minimal benefit, transient improvement or variability in response between individuals.
A clinically responsible framework therefore recognises several principles supported by microbiome research:
- Substantial variation: there is substantial variation in microbial composition between individuals, even among healthy population
- Functional differences at species and strain level that influence metabolic activity
- Context-dependent metabolic effects rather than outcomes determined by the presence of a single organism
- Bidirectional signalling between host physiology and microbial activity 3,4,16,17
Key Takeaway
The gut microbiome is a measurable, modifiable part of human physiology. It functions as an ecosystem — and like any ecosystem, its health depends on the balance, diversity, and functional capacity of the community as a whole, not just the presence or absence of individual organisms.
For clinicians, the most useful insight from the microbiome is often functional rather than taxonomic: what the microbial community is producing, how that interacts with the patient’s immune system, gut barrier, and metabolic regulation, and how those outputs may be shifting in the context of that individual’s diet, medications, and clinical picture.
For clinicians, microbiome insights provide an additional lens for understanding systemic regulation, particularly in chronic and complex symptom patterns.
As our understanding of the microbiome deepens, so does the opportunity to use it as a meaningful part of clinical assessment, particularly for patients with chronic or multi-system presentations where traditional markers tell only part of the story.

1. Fan Y, Pedersen O. Gut microbiota in human metabolic health and disease. Nat Rev Microbiol. 2021;19(1):55–71. https://doi.org/10.1038/s41579-020-0433-9
2. Lynch SV, Pedersen O. The human intestinal microbiome in health and disease. N Engl J Med. 2016;375(24):2369–2379. https://doi.org/10.1056/NEJMra1600266
3. Wilde J, Slack E, Foster KR. Host control of the microbiome: Mechanisms, evolution, and disease. Science. 2024;385(6706):eadi3338. https://doi.org/10.1126/science.adi3338
4. Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012;486(7402):207–14.
5. Mann ER, Lam YK, Uhlig HH. Short-chain fatty acids: linking diet, the microbiome and immunity. Nat Rev Immunol. 2024;24(8):577–595. https://doi.org/10.1038/s41577-024-01014-8
6. de Vos WM, Tilg H, Van Hul M, Cani PD. Gut microbiome and health: mechanistic insights. Gut. 2022;71(5):1020–1032. https://doi.org/10.1136/gutjnl-2021-326789
7. Zhou X, Shen X, Johnson JS, et al. Longitudinal profiling of the microbiome at four body sites reveals core stability and individualized dynamics during health and disease. Cell Host Microbe. 2024;32(4):506–526.e9.
8. Hays KE, Pfaffinger JM, Ryznar R. The interplay between gut microbiota, short-chain fatty acids, and implications for host health and disease. Gut Microbes. 2024;16(1):2393270. https://doi.org/10.1080/19490976.2024.2393270
9. Ornelas A, Dowdell AS, Lee JS, Colgan SP. Microbial Metabolite Regulation of Epithelial Cell-Cell Interactions and Barrier Function. Cells. 2022;11(6):944.
10. Honda K, Littman DR. The microbiota in adaptive immune homeostasis and disease. Nature. 2016;535(7610):75–84. https://doi.org/10.1038/nature18848
11. Koh A, De Vadder F, Kovatcheva-Datchary P, Bäckhed F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell. 2016;165(6):1332–1345. https://doi.org/10.1016/j.cell.2016.05.041
12. Muramatsu MK, Winter SE. Nutrient acquisition strategies by gut microbes. Cell Host Microbe. 2024;32(6):863–874. https://doi.org/10.1016/j.chom.2024.05.011
13. Cryan JF, O’Riordan KJ, Cowan CSM, et al. The microbiota-gut-brain axis. Physiol Rev. 2019;99(4):1877–2013. https://doi.org/10.1152/physrev.00018.2018
14. Lloyd-Price J, Arze C, Ananthakrishnan AN et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature. 2019;569(7758):655–662. https://doi.org/10.1038/s41586-019-1237-9
15. Staudacher HM, Mikocka-Walus A, Ford AC. Common mental disorders in irritable bowel syndrome: pathophysiology, management, and considerations for future randomised controlled trials. Lancet Gastroenterol Hepatol. 2021;6(5):401–410. https://doi.org/10.1016/S2468-1253(20)30363-0
16. Leshem A, Segal E, Elinav E. The Gut Microbiome and Individual-Specific Responses to Diet. mSystems. 2020;5(5):e00665-20.
17. Zmora N, Suez J, Elinav E. You are what you eat: diet, health and the gut microbiota. Nat Rev Gastroenterol Hepatol. 2019;16(1):35–56. https://doi.org/10.1038/s41575-018-0061-2